|
Extensive ore reserves are found in Canada, Brazil, Nigeria, Zaire, and in Russia. Large deposits of niobium have been found associated with carbonatites (carbon-silicate rocks), as a constituent of parochlore. The element is found in niobite (or columbite), niobite-tantalite, parochlore, and euxenite. commercial producers, however, still refer to the metal as "columbium." Sources Most metallurgists, leading metal societies, and all but one of the leading U.S. Many leading chemical societies and government organizations refer to it by this name. The name niobium was adopted by the International Union of Pure and Applied Chemicstry (IUPAC) in 1950 after 100 years of controversy. The metal was first prepared in 1864 by Blomstrand, who reduced the chloride by heating it in a hydrogen atmosphere. Discovered in 1801 by Hatchett in an ore sent to England. Named after Niobe, the daughter of Tantalu. Niobium alloy steels are often used in pipelines.ĥ, 4, 3, 2, 1, −1, −3 (a mildly acidic oxide) This is clearly shown in the figure of the orbital diagram of niobium.Niobium metal is used in jewelry and as an alloying agent in steel to increase its strength. So an electron will enter the 5s orbital in the clockwise direction and the remaining four electrons enter the 4d orbital in the clockwise direction. So, the next six electrons will enter the 4p orbital just like the 3p orbital. Therefore, the next five electrons will enter the 3d orbital in the clockwise direction and the next five electrons will enter the 3d orbital in the anti-clockwise direction. So, the next two electrons will enter the 4s orbital just like the 1s orbital. Then the next three electrons will enter the 3p orbital in the clockwise direction and the next three electrons will enter the 3p orbital in the anti-clockwise direction. Then the next two electrons will enter the 3s orbital just like the 1s orbital. The next three electrons will enter the 2p orbital in the clockwise direction and the next three electrons will enter the 2p orbital in the anti-clockwise direction. The next two electrons will enter the 2s orbital just like the 1s orbital. The 1s orbital is now filled with two electrons. Therefore, the electrons will first enter the 1s orbital.Īccording to Hund’s principle, the first electron will enter 1s orbital in the clockwise direction and the next electron will enter the 1s orbital in the anti-clockwise direction. When electrons want to enter the p-subshell, then the first electron will enter the p x orbital in the clockwise direction.ġs is the closest and lowest energy orbital to the nucleus. The orbitals are p x, p y, and p z and each orbital can have a maximum of two electrons. Hund’s principle is that electrons in different orbitals with the same energy would be positioned in such a way that they could be in the unpaired state of maximum number and the spin of the unpaired electrons will be one-way.įor example, we already know that the p-subshell has three orbitals. Hund’s principle is a rule that helps to determine how electrons are distributed in orbitals when multiple orbitals of the same energy level are available. Now it is possible to find the orbital notation of niobium very easily through electron configuration. The atomic number of niobium is 41, which means it has 41 electrons. The Aufbau principle is that the electrons present in the atom will first complete the lowest energy orbital and then gradually continue to complete the higher energy orbital. This orbital notation system always follows the Aufbau principle. This is similar to electron configuration, but numbers are used instead of boxes to represent the positions of the electrons. The niobium orbital notation is a shorthand system designed to represent the exact positions of the electrons in the niobium atom. What is the orbital notation for niobium? The sub-energy level ‘s’ can hold a maximum of two electrons, ‘p’ can hold a maximum of six electrons, ‘d’ can hold a maximum of ten electrons, and ‘f’ can hold a maximum of fourteen electrons. Each orbital can have a maximum of two electrons. The orbital number of the s-subshell is one, three in the p-subshell, five in the d-subshell, and seven in the f-subshell. The sub-energy levels are known as s, p, d, and f. The sub-energy levels depend on the azimuthal quantum number. 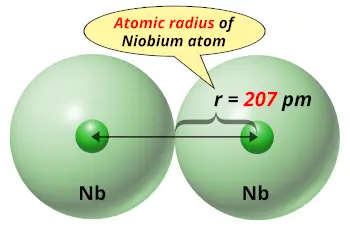
The most probable region of electron rotation around the nucleus is called the orbital. These sub-energy levels are also called orbital. Again, atomic energy shells are subdivided into sub-energy levels. These circular paths are called orbit (shell). The electrons of the atom revolve around the nucleus in a certain circular path.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
